 Another silicate of magnesium is talc, Mg 3Si 3O 10(OH) 3. Another fibrous magnesium silicate without iron is chrysotile. Those minerals include tremolite, riebeckite and grunerite. Magnesium, along with iron, is a constituent of the fibrous silicate minerals which were collectively given the commercial name asbestos. Magnesium appears with zinc and arsenic in the silicate mcgovernite. Magnesium appears in the carbonate mineral Benstonite. Magnesium forms the hydroxide Mg(OH) 2 which in mineral form is called Brucite. It forms an oxide along with manganese and other metals in the mineral birnessite. With aluminum it forms the oxide mineral spinel. Another oxide with boron (borate) with iron and titanium is warwickite. The compound with boron is called boracite. It appears with chromium in the carbonate mineral stichtite. It appears with copper in the carbonate mineral Callaghanite. Magnesium is found with boron in the oxide minerals inderite and kurnakovite. It appears with boron in the fibrous mineral Szaibelyite. It appears with iron in the carbonate pyroaurite and in the mineral Hulsite. Magnesium is contained in the carbonate minerals dolomite, CaMg(CO 3) 3, magnesite MgCO 3 and artenite Mg 2CO 3(OH) 2.3H 2O. Magnesium is the final member of the big 8 elements in the Earth's crust, being the eighth most abundant element at about 2.1% by weight. Magnesium is a key component in chlorophyll, the molecule in green plants which helps transform sunlight and carbon dioxide into water and sugar. Magnesium sulfate, MgSO 4.7H 2O is called Epsom salt and is used as a cleansing agent. Magnesium oxide, MgO is called magnesia and is used medically for neutralizing excess stomach acid and as a laxative ("milk of magnesia" is MgO in water). Magnesium powder and an oxidizing agent is used as flash powder. The metal burns in air with a bright white light, leading to application in flares. Magnesium reacts with boiling water to form magnesium hydroxide and hydrogen gas. This low density is used to advantage in alloys like magnalium (10% magnesium, 90% aluminum). Only calcium and the alkali metals are lighter. Molecular weight of Magnesium Sulfate heptahydrate (MgSO 4.Magnesium is a very light metal. Magnesium Sulfate heptahydrate Molecular Weight Calculation Molecular weight of Magnesium Sulfate heptahydrate (MgSO 4.7H 2O) So the molecular weight of Magnesium Sulfate heptahydrate is 246.4764. Step 4: Calculate the molecular weight of Magnesium Sulfate heptahydrate by adding up the total weight of all atoms. Total weight of water in Potassium antimonyl tartrate trihydrate: 18.0154 x 7 = 126.1078 
Number of water (H 2O) molecules in Potassium antimonyl tartrate trihydrate: 7 Total weight of Oxygen atoms in Magnesium Sulfate heptahydrate: 15.9994 x 4 = 63.9976 Number of Oxygen (O) atoms in Magnesium Sulfate heptahydrate: 4 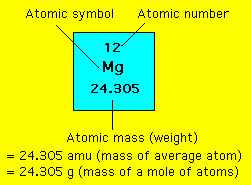
Total weight of Sulfur atoms in Magnesium Sulfate heptahydrate: 32.066 x 1 = 32.066 Number of Sulfur (S) atoms in Magnesium Sulfate heptahydrate: 1 Total weight of Magnesium atoms in Magnesium Sulfate heptahydrate: 24.305 x 1 = 24.305 Number of Magnesium atoms in Magnesium Sulfate heptahydrate: 1 Step 3: Calculate the molecular weight of Magnesium Sulfate heptahydrate by adding the total weight of all atoms. Step 2: Find out the atomic weights of each atom (from the periodic table).Ītomic weight of Magnesium (Mg) : 24.305 (Ref: Jlab-ele012)Ītomic weight of Sulfur (S) : 32.066 (Ref: Jlab-ele016)Ītomic weight of Oxygen (O): 15.9994 (Ref: Jlab-ele008) From the chemical formula, you can find that one molecule of Magnesium Sulfate heptahydrate has one Magnesium (Mg) atom, one Sulfur (S) atom, four Oxygen (O) atoms, and seven water molecules. 
The chemical formula of Magnesium Sulfate heptahydrate is MgSO 4.7H 2O. You will know different atoms and their number in a Magnesium Sulfate heptahydrate molecule from the chemical formula. Step 1: Find out the chemical formula and determine constituent atoms and their number in a Magnesium Sulfate heptahydrate molecule. CALCULATION PROCEDURE: Magnesium Sulfate heptahydrate Molecular Weight Calculation The molecular weight of Magnesium Sulfate heptahydrate is 246.4764 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements and water molecules. The heptahydrate form of Magnesium sulfate (MgSO 4.7H 2O, also known as Epsom Salt) also contains 7 water molecules. Magnesium Sulfate (MgSO 4) is an inorganic compound of three elements: Magnesium, Sulfur, and Oxygen. The molecular weight of Magnesium Sulfate heptahydrate (MgSO 4.7H 2O) is 246.4764. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
